How to Use ChemCalc App - A chemistry calculator and study guide game
To use ChemCalc, start by downloading the Android app to your Android smartphone.
Link to ChemCalc for Android smartphones
for Android smartphones
ChemCalc is both a calculator and a periodic table of elements.
Use ChemCalc to do chemistry calculations or to look up information about elements in the full periodic table of 118 elements.
Students and teachers can use ChemCalc to calculate the molar mass or molecular weight of molecules.
For example to get the molar mass of water:
H2O
you can tap on:
( H * 2 ) + O =
Or to get the molar mass of carbon dioxide:
CO2
you can tap on:
C + ( O * 2 ) =
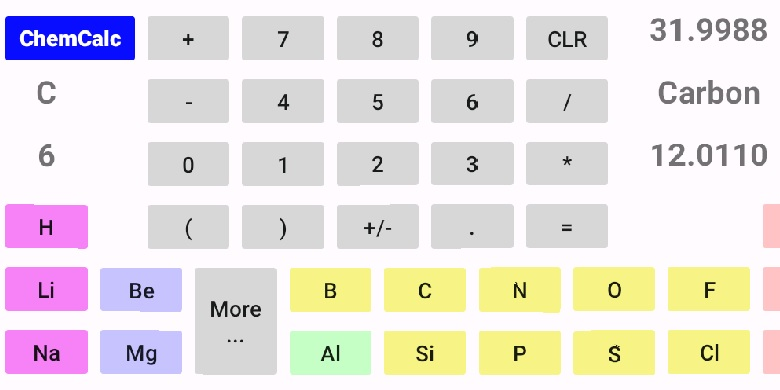
As you tap on the elements symbol, the elements name, atomic number, and atomic weight are displayed. If you want more information about an element, hold down on the elements symbol instead of a quick tap. If you are interested in an element not on the calculator screen, select the 'More ...' button or just swipe up on the calculator screen. This will display the remainder of the periodic table that you can scroll left or right to locate the element you're trying to find.
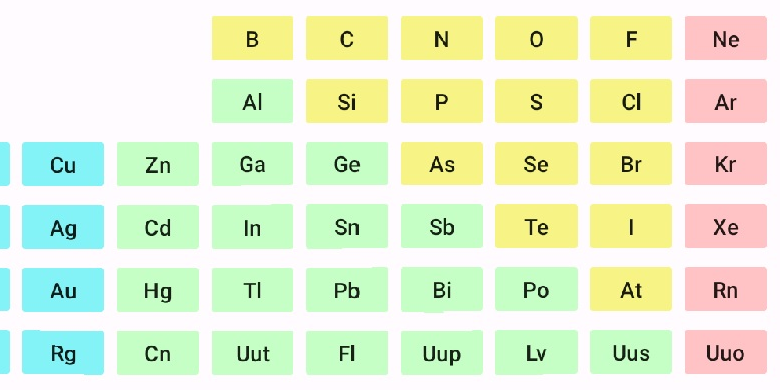
ChemCalc also includes a set of flash cards for students studying the elements called ChemCards, and a game for students memorizing the order of elements in the periodic table called ChemGame. Also included is ChemGame which will test students memory of the periodic table. In ChemGame you get points for matching elements that are next to each other horizontally or vertically in the periodic table. More points are awarded for elements next to each other vertically than horizontally, and farther down the periodic table. Compete with classmates to see who can get the highest score.
As mentioned in other pages on this web site, plants use energy from sunlight to chemically change water (H2O) and carbon dioxide (CO2) into sugar. This chemical process is described by the following chemical equation:
- 6(CO2) + 6(H2O) + light -> C6H12O6 + 6(O2)
where:
- CO2 is carbon dioxide
- H2O is water
- C6H12O6 is sugar
- O2 is oxygen
Also, noted on other pages on this web site, launch systems typically use chemical rockets to accelerate spacecraft to Earth orbit or farther. Rockets use fuels that go through a chemical reaction to release energy. This released energy heats the exhaust gas creating a lot of pressure in the rocket engine combustion chamger. This pressurized gas is guided by the rocket engine nozzle into a high-speed exhaust stream which causes the rocket acceleration. Some common rocket fuels are:
- liquid propellants
- Liquid hydrogen and liquid oxygen (LH2/LOX) 2(H2) + O2 -> heat + 2(H2O)
- Kerosene and liquid oxygen (RP-1/LOX)
- Unsymmetrical dimethylhydrazine and dinitrogen tetroxide (UDMH/N2O4) N2C6H8 + N2O4
- Aerozine/dinitrogen tetroxide (A-50/N2O4)
- Ammonium perchlorate composite solid propellants (APCP)
- aluminium and ammonium perchlorate Al + NH4ClO4
- magnesium and ammonium perchlorate Mg + NH4ClO4
- zinc and ammonium perchlorate Zn + NH4ClO4
- Hydroxyl-terminated polybutadiene (HTPB) and ammonium perchlorate
- Polybutadiene acrylonitrile (PBAN) and ammonium perchlorate
This chemistry calculator requires no network connection allowing you to work offline and use it anywhere.
Link to ChemCalc for Android smartphones
for Android smartphones
Copyright (C) 2017 R. J. Kuhn. Please note that you are not allowed to reproduce or rehost this page without written permission.

